|
1 – 5, 7, 11, 23 – 26 From a practical perspective, specific vehicles might be more applicable to certain body sites due to specific factors, such as ease of application, spreadability, emolliency, lack of residue, and ease of wash-off. 23, 26 Limitations of use include general avoidance on certain body locations (e.g., face, inguinal/genital region), necessary application to an extensive BSA, especially over prolonged durations of therapy, and use of Class I TCs in children. 23, 26Ĭlass I TCs are superior to Class II TCs in achieving clearance or near-clearance of psoriatic plaques. 1 – 5, 7, 11, 23 – 26 The literature supports the use of primarily super-potency (Class I) or high potency (Class II) TCs for the treatment of plaque psoriasis, especially to achieve adequate control of a disease flare or persistent psoriatic lesions. Clinicians often involve TCs early in the management of plaque psoriasis, especially when limited body surface area (BSA) is affected however, TCs are used for all severities of disease, and several factors-such as patient age, affected anatomic locations, extent of BSA involvement, prior therapies utilized, and other concurrent treatments-affect the potency of agents and the vehicles that are selected. TCs IN THE MANAGEMENT OF PLAQUE PSORIASIS In this report, the author provides an overview of managing plaque psoriasis with TCs, discusses product selection tendencies, addresses conventional use and formulations of topical clobetasol propionate (CP) 0.05%, reviews the use of special additives (e.g., penetration enhancers) to increase CP potency, and highlights results from a recent study on a new CP cream formulation. 1 – 5, 7, 9 – 16 It cannot be assumed that generic formulations containing the same concentration of active ingredient are equivalent to brand formulations that incorporate a different vehicle composition and/or formulation technology, as PK effects, clinical efficacy, skin tolerability, and patient satisfaction and adherence might differ substantially between the two. 
The formulation determines the pharmacokinetic (PK) properties of the TC product (e.g., skin penetration, active ingredient release/skin delivery, skin tolerability), and the vehicle characteristics affect whether the patients like the feel of the product (i.e., cosmetic acceptability) and will adhere with application instructions. In addition to the potency and anticipated efficacy of a given TC, vehicle formulation and patient preference are major factors that directly influence the success or failure of TC therapy. Less often, or especially with lower-potency TCs, some pivotal trials are designed to test the medication in subjects with AD that include adolescent and pediatric age groups. 8 As a result, newer and more potent TC formulations are usually studied in adult subjects with plaque psoriasis. The FDA-approved indication of a novel TC formulation now specifies the disease state and age group for which it is indicated (e.g., “topical treatment of plaque psoriasis in patients 18 years of age and older”). 1, 5, 7 Nevertheless, the FDA has more recently disbanded the use of the broad CRD category and now restricts disease-state approval to the specific skin disorder evaluated in the pivotal large-scale, randomized, controlled clinical trials (RCTs) that are required during formal product development prior to submission to the FDA for approval. 1, 4, 5 The diverse biologic properties of TCs appear to contribute directly to their positive therapeutic effects in many skin disorders these effects include anti-inflammatory properties, stabilization of cellular and lysosomal membranes, reduction in neutrophil and monocyte recruitment, decreased lymphocyte reactivity, modulation of Langerhans cell activity/expression, antiproliferative/antimitotic effects, vasoconstrictive properties, and reduction in mast cell density and reactivity, including immunoglobulin E sensitization. 6 The list of CRDs could include psoriasis, AD, allergic contact dermatitis, irritant contact dermatitis, dyshidrotic eczema, lichen planus, cutaneous lupus, granuloma annulare, and others. 1 – 5 For several years, when highlighting the presence of an indication approved by the United States (US) Food and Drug Administration (FDA) for a TC brought to the US marketplace, product labeling has included the following description: “topical corticosteroids are indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses” (CRDs). 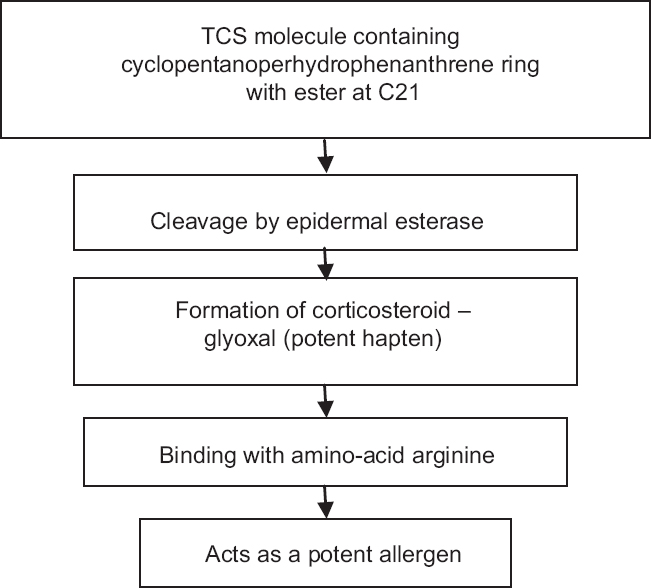 
Ever since topical hydrocortisone was first introduced, in 1952, as a treatment for eczematous dermatitis, topical corticosteroids (TCs) have maintained a central position in the therapeutic armamentarium for several dermatologic disorders, including psoriasis, atopic dermatitis (AD), contact dermatitis, seborrheic dermatitis, and other inflammatory dermatoses.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
